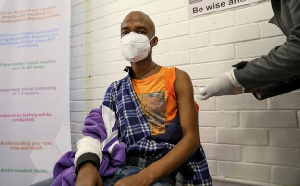
The African Union Commission has launched a consortium for Covid-19 vaccine clinical trials aimed at securing more than 10 trials as early as possible.
The new consortium will be headed by the Africa Centres for Disease Control and Prevention.
It will bring together global vaccine developers, funders, and African organisations that facilitate clinical trials to ensure that sufficient data on effects of vaccine candidates on the African population is generated. This will enable vaccines to be confidently rolled out in Africa once approved.
The AU Commission says rolling out a vaccine in Africa is key to “limiting transmission, preventing deaths and laying the foundation for socio-economic recovery that should bring Africa’s integration agenda back on track”.
“Success in developing and providing access to a safe vaccine requires an innovative and collaborative approach, with significant local manufacturing in Africa. We need to support the contribution of African scientists and healthcare professionals. We need to act with urgency,” said AU Chairperson President Cyril Ramaphosa.
South Africa and Egypt are already running human trials for a potential vaccine.
- Expanding Africa’s vaccine production capacity key – Noguchi Director
- Dengue fever epidemic declared in Burkina Faso
- Frontiers earned over $87m while government got under $7m from COVID testing at KIA – Report
- I donated PPE worth over $1 million during COVID-19 pandemic – Ken Agyapong
- Ablakwa releases ‘inaccessible details’ of contract awarded to Frontiers for COVID test at the airport
- Read all related articles